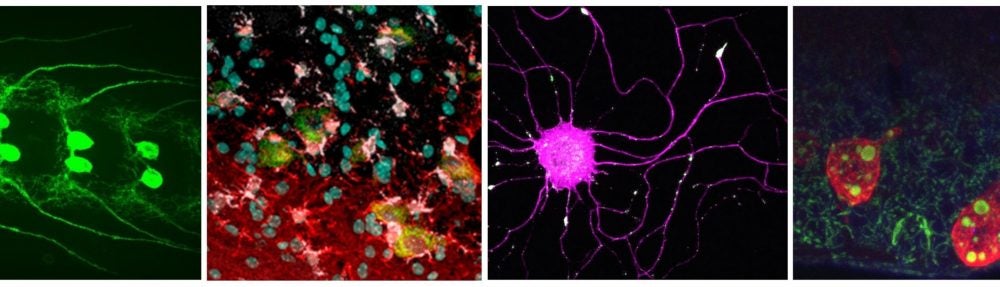
Axons connect single neurons to distant parts of the brain and body. During development, the directional growth and branching of axons underlies the formation of neuronal circuitry, and in adults, the structure and integrity of axons is essential for proper function and maintenance of the nervous system.
A challenge to treating injuries to the CNS, including spinal cord injuries, neurodegenerative diseases, and stroke, is that we have few methods to stimulate and control the re-growth and/or new growth of axons. Despite the presence of inhibitory factors to regeneration in the CNS, most neurons have an intrinsic capacity to initiate axonal re-growth after injury. We are interested in the cellular processes that underlie the initiation of regeneration in neurons.
We have developed a new assay that allows for the study of regeneration and injury responses in Drosophila larvae. A highly conserved axonal kinase, Wallenda, (DLK in vertebrates) is required in neurons to initiate a regenerative response to axonal injury.
 TheGal4/UAS system is used to label single axons in larval segmental nerves, which have been subjected to nerve crush. We can use this assay to study regenerative responses to injury in live, genetically tractable, animals . (From Xiong et al., (2010) Journal of Cell Biology 191:211-23). PubMed
TheGal4/UAS system is used to label single axons in larval segmental nerves, which have been subjected to nerve crush. We can use this assay to study regenerative responses to injury in live, genetically tractable, animals . (From Xiong et al., (2010) Journal of Cell Biology 191:211-23). PubMed
We are interested in the following questions:
1. How does a neuron ‘know’ that it has been injured?
The Wnd kinase regulates a nuclear signaling cascade that induces dramatic changes in gene expression in the neuron after injury. Studies in vertebrates suggest that specific signaling molecules are transported in axons, and that this is necessary for the cell body to receive information from distant sites in the axon. Wnd is transported in axons, and intriguingly, axonal transport machinery is required for injury signaling. We’re taking a combined approach of genetics and biochemistry to understand the mechanism of injury signaling on a molecular level.
We are also studying the mechanism by which the Wnd kinase becomes activated in axons. Combined observations in Drosophila, C. elegans, and vertebrate neurons suggest that this kinase functions as an injury ‘sensor’. Local protein turnover in axons is an important element of its regulation and its activation.
2. What is the response to injury on a molecular level?
The Wnd-regulated signaling pathway, which is required for initiation of a regenerative response, is an important inroad into identifying the intrinsic injury responses in neurons that are permissive for regeneration. We are also interested in the role that surrounding glia play, and the cellular mechanisms that underlie glial responses to axonal injury.
3. How do axons degenerate after injury?
After injury, the part of the axon which is no longer attached to the neuron degenerates through a regulated but poorly understood process called Wallerian degeneration. Our injury model allows for us to use powerful genetics and imaging techniques to study this process at the Drosophila NMJ. We are especially interested in the cellular events that initiate this process, and their relationship to events that occur in neuropathies and degenerative disorders that involve degeneration of axons.
4. What is the relationship of synaptic plasticity and regeneration?
Injury interrupts synaptic connections, and regeneration aims to restore synaptic connections. Are molecular pathways used for regeneration also used for synaptic plasticity, and vice versa?
Live Imaging
Important elements of our approaches in the lab include Drosophila genetics, biochemistry, and live imaging. We using a spinning disk confocal microscope to study many cellular aspects of neurons in living Drosophilaanimals, and are currently developing new methods to image axonal and glial responses to injury. Here are some example movies
Axonal transport
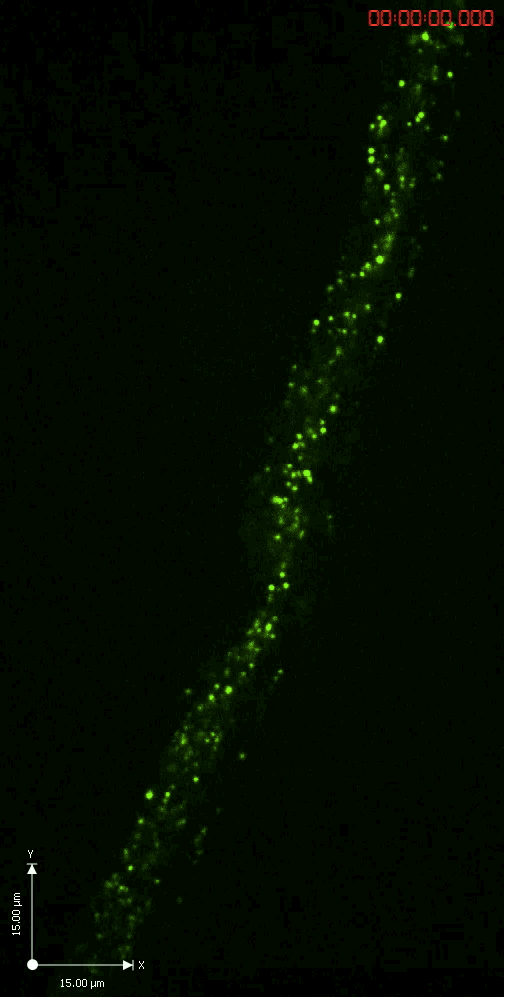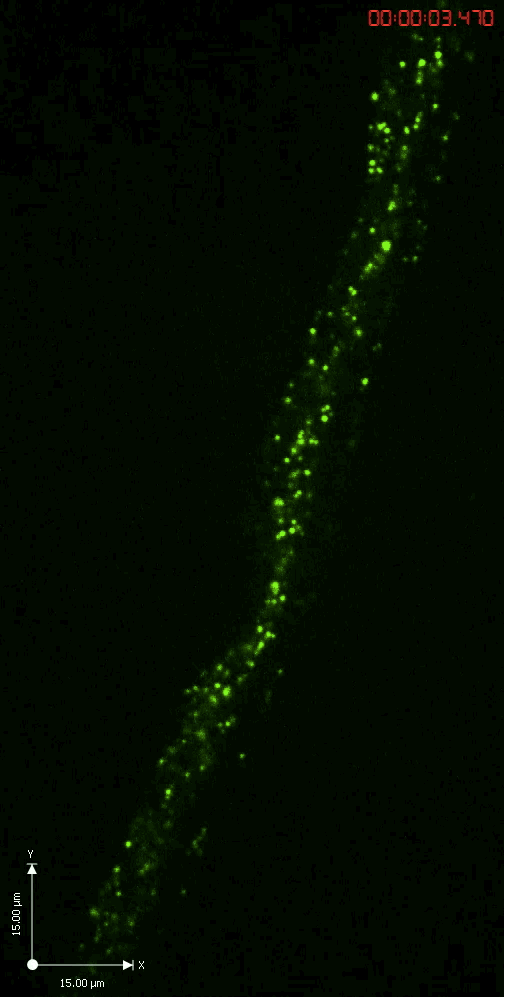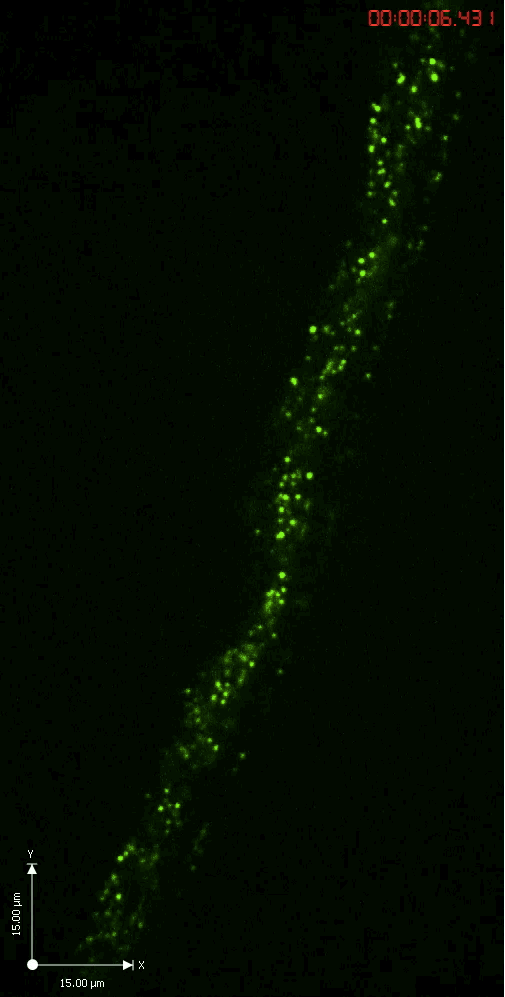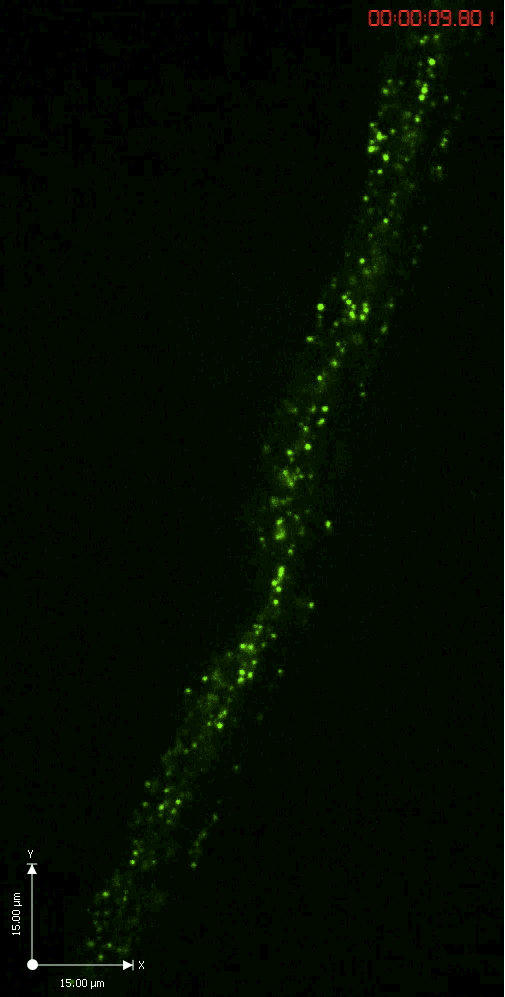
A GFP-tagged synaptic vesicle cargo in Drosophila motoneurons. What you see is rapid time lapse imaging of axonal transport in segmental nerves of a Drosophila larva. The anterograde (upward moving) and retrograde (downward moving) vesicles are carried by kinesin and dynein motor proteins within individual axons in the nerve.
Calcium imaging in response to injury

A dendrite of a sensory neuron is cut with a pulsed UV laser. The GCamP3 calcium sensor detects rapid local increases in calcium in the dendrite, which quickly translates to a large calcium increase in the cell body. To immobilize animals we are collaborating with Nikos Chronis’s lab in mechanical engineering, in the application of a microfluidics device for live imaging in Drosophila.