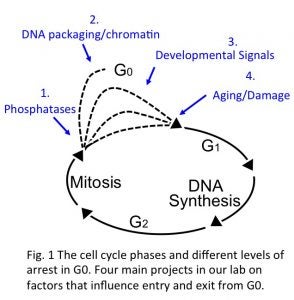
The cell cycle is an ordered series of molecular events in a cell leading to division and production of two daughter cells. This process is fundamental to the development of all multicellular organisms, which begin from a single cell, the fertilized egg. Equally important for proper development though, is the slowing or stopping of the cell cycle at the right places and times as cells, tissues and organs mature. In fact, the majority of cells in multicellular organisms spend most of their existence in non-proliferating states, often referred to as cellular quiescence or the G0 phase (Fig.1).
In their non-dividing state, quiescent cells are metabolically active and carry out critical physiological functions in tissues and organs. Despite the importance of G0, most studies of cell cycle regulation have focused on rapidly dividing cells. We thus understand very little about why or how cells choose to enter quiescence during development, and why some cells can choose to leave G0 and later re-enter the cell cycle in response to developmental signals, tissue damage, or nutrient availability. We are now beginning to recognize that there are multiple states of quiescence. Some quiescent states are readily reversible, as in the cases of resting stem cells or nutrient starved cells, while others are permanent, such as the G0 associated with the final stages of tissue development for example, in muscles and neurons. It is becoming clear that different quiescent states are controlled in distinct ways, both in the manner of cell cycle inhibition and in the pathways used to initiate exit.
The goal of our research is to understand how the cell cycle machinery is controlled during the decision to exit the cell cycle, and how quiescence (G0) can be modulated to be more or less reversible in different contexts. To accomplish this we use the combination of a genetically tractable model organism, Drosophila melanogaster (fruit fly) for detailed in vivo genetic manipulations of cell cycle regulators during development and mammalian (mouse and human) cell lines to examine the evolutionary conservation of the pathways we discover and their potential relevance to cancer.
Our research impacts a wide range of biological questions, as the proper control of quiescence is critical in development and tissue regeneration, but becomes disrupted in aging and cancer.
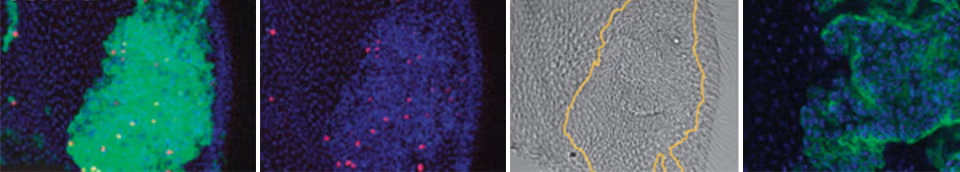
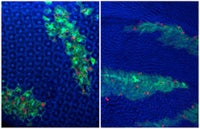 Photo: Differentiated post-mitotic cells in the fly eye (left) and wing (right) can be induced to re-enter the cell cycle and undergo mitoses (red) by the expression of a few defined cell cycle factors (expressing cells labeled with GFP).
Photo: Differentiated post-mitotic cells in the fly eye (left) and wing (right) can be induced to re-enter the cell cycle and undergo mitoses (red) by the expression of a few defined cell cycle factors (expressing cells labeled with GFP).
Like our science images? See more at Bioartography.com
Contact Dr. Buttitta
Email | buttitta@umich.edu
Office | (734) 764-2802
5218 Biological Sciences Bldg.
Lab | (734) 936-7277
5224 Biological Sciences Bldg.