Our Research
Lipid bilayer membranes containing cholesterol can support two coexisting liquid phases, called liquid-ordered and liquid-disordered. There is evidence that the presence of this phase transition impacts the organization and function of plasma membrane proteins. We are currently exploring the physical basis and functional consequences of lateral heterogeneity in living cell membranes using a variety of experimental and theoretical approaches, including super-resolution fluorescence localization microscopy and Monte Carlo modeling of basic signaling processes.
IMMUNE CELL SIGNALING
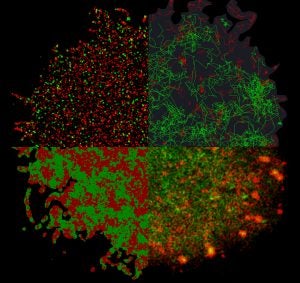
Recognition of antigen by the B cell receptor is crucial to the adaptive immune response and, ultimately, defending the body from foreign pathogens. B cell receptors become activated when clustered by pathogens or artificial crosslinkers and initiate a cellular-level response. Initial B cell receptor activation is regulated by a large number of proteins at the cell surface. We explore the spatial organization proteins in the plasma membrane using multi-color super-resolution microscopy.
ION CHANNEL REGULATION
Ion channels are proteins that regulate the flow of specific ions across membranes. These membrane proteins sense and transmit information in neurons and are often sensitive to hydrophobic small molecules that also alter the size and stability of membrane domains. We are investigating if and how membrane domains couple to certain ion channels in model and neuronal systems.

COUPLED PHASE TRANSITIONS
Liquid droplets found in the cytoplasm and nucleus of cells assemble through a mechanism that is distinct from the liquid-liquid phase transition observed in membranes. We are interested in how these polymer phase transitions can couple to membrane phase transitions to drive novel cellular functions.
SUPER-RESOLUTION LOCALIZATION MICROSCOPY
We acquire fluorescence localization data in chemically fixed and live cells and develop analytical methods to extract useful information from super-resolution fluorescence localization data-sets.