Project Goals and Current Work
The primary goal on the Manganese Project is to understand the role of manganese in biological systems more completely. Specifically we are interested in the redox active manganese-containing enzymes, with an emphasis on catalase, manganese superoxide dismutase, and the oxygen evolving complex. Our approach to these systems has been to prepare model complexes which are then characterized and explored with respect to their abilities to act as functional, structural or spectroscopic models for the enzyme in question. The following paragraphs provide a brief introduction to the three enzymes mentioned above and then describe some of the exciting results that have come out of this laboratory during our explorations of the bioinorganic chemistry of manganese. The results discussed have been published and references are provided. Work on all of these projects is ongoing.
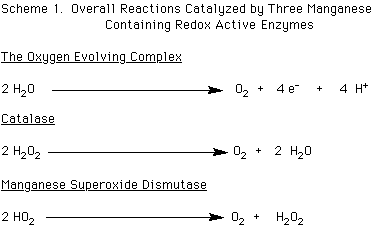
The oxygen evolving complex, OEC, is part of photosystem II, PSII. This enzyme catalyzes the oxidation of two molecules of water to yield one molecule of dioxygen, four protons, and four equivalents of electrons, as shown in Scheme 1 below.
The OEC is part of the overall photosynthetic system that harnesses light energy for the eventual reduction of carbon dioxide and water to form sugars in green plants and algae. The core of the OEC contains a cluster of four manganese ions. The OEC is successively oxidized in four one electron steps to yield four equivalents of electrons that are used as reducing equivalents. Most, if not all, of these oxidations occur on the tetranuclear core of manganese ions. After the fourth oxidation, a molecule of dioxygen is released, and the OEC returns to a lower overall oxidation state. This cycle was established by studying the periodic bursts of dioxygen that occurred with every fourth flash of light given to preparations of photosynthetic centers by Kok in the early 1970’s, and has been named the S-state cycle, which proceeds from S0 to S4. Each electron drawn from the OEC is transferred via a redox active tyrosine residue, know as tyrosine Z, to an oxidized chlorophyll molecule that was excited and oxidized by absorbed light energy. The electron that had been lost from the chlorophyll then goes on to be utilized as a reducing equivalent further along the line in the photosynthetic system. (Debus, R. J., Biophys. et. Biochim. Acta, 1992, 1102, 269; Oxygenic Photosynthesis: The Light Reactions, Ort, D. R. and Yocum, C. F., Eds.; Kluwer Academic Publishers: 1996)
Catalase contains two manganese ions at its active site, while manganese superoxide dismutase, MnSOD, is a mononuclear enzyme. Both of these enzymes play a role in protecting an organism from the potential deleterious effects of the reduced forms of dioxygen, hydrogen peroxide and superoxide, respectively. While catalase to date has been found predominately in bacteria, manganese superoxide dismutase is more widespread, being found in a range of organisms from bacteria to the mitochondria of humans. Catalase catalyzes the disproportionation of two hydrogen peroxide molecules to two molecules of water and one of dioxygen. MnSOD catalyzes the dismutation of two molecules of superoxide of one molecule of hydrogen peroxide and one of dioxygen. The catalase enzyme completes its catalytic cycle in two two-electron reduction and oxidation steps, while MnSOD engages in a one electron process. The overall processes are depicted in Scheme 1 above. (Pecoraro, V. L. et al. Chem. Rev., 1994, 94, 807; Manganese Redox Enzymes, Pecoraro, V. L., Ed.; VCH Publishers, New York: 1992).
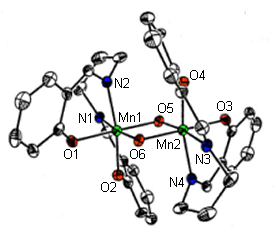
One class of molecules that we have explored are based on the ligand H2salpn, N,N’-bis(salicylidene)-1,3-diaminopropane. With this ligand and derivatives thereof, we have prepared a family of manganese dimers in the Mn(IV)Mn(IV) oxidation state with two bridging �-O2-‘s to yield Mn2O2 cores (Larson, E. J., and Pecoraro, V. L. J. Am. Chem. Soc., 1991, 112, 3810). A crystal structure of the underivatized dimer, [Mn(IV)(salpn)( �-O)]2 appears in the link to this page, and is also shown in figure 1 below.
This [Mn(IV)(salpn)( �-O)]2 complex structurally resembles a part of the OEC Mn-assembly, as it has a Mn-Mn separation of 2.7 , precisely the distance deduced for the Mn-Mn separation in the lower S-states of the enzyme. This dimer has proven particularly useful for our studies of manganese chemistry related to the function of the OEC. In 1991, our group reported that this complex is a very competent catalase mimic, and catalytically disproportionates hydrogen peroxide (Larson, E. J., and Pecoraro, V. L. J. Am. Chem. Soc., 1991, 112, 7809). This is interesting in light of the possible centered catalase-like reaction of the OEC. Another key aspect of this system has been our ability to protonate the oxo-bridges of the compound, and consequently to probe changes in the physical properties of this system. Our data has shown that a broad range of pKa’s for the protonation of the oxo-bridges exists for this family of complexes and that there is also a broad range of reduction potentials, which indicates how nature may easily be able to control the potential at the tetranuclear manganese cluster of the OEC (Baldwin, M. J., et al., Photosynth. Research, 1993, 38, 303). Magnetic susceptibility studies have shown that successive protonation of the two oxo-bridges reduces the coupling of the two manganese centers to one another. Furthermore, these protonations lead to a lengthening of the Mn-Mn vector in the complex (Baldwin, M. J., et al., J. Am. Chem. Soc., 1994, 115, 11349).
These results may have implications for the functioning of the OEC. New proposals regarding the oxidation of water to dioxygen now suggest that the successive oxidations of the OEC may occur via a hydrogen atom abstraction process, thus coupling the removal of an electron with a concurrent loss of a proton, possibly from a water bound to one of the manganese ions in the OEC. Based on recent studies of tyrosine Z, it has been suggest that the H-atom abstraction is conducted by tryosine Z, the electron transfer agent between the OEC and the chlorophyll. We have utilized our system of [Mn(IV)(salpn)( µ-O)]2 complexes to explore the feasibility of the H-atom abstraction proposal. To do that, we utilized the properties of this system to calculate the homolytic bond dissociation energy, HBDE, for the loss of an H-atom from the protonated oxo-bridges of these complexes. Our data suggest that such a process is feasible (Baldwin, M. J. and Pecoraro, V. L. J. Am. Chem. Soc., 1996, 118, 11325-11326).
A second class of dimeric compounds that has provided many exciting results is based on the ligand H3(2-OHsalpn), N,N’-bis(salicylidene)-2-hydroxy-1,3-diaminopropane. This system is unique in many ways and has allowed us to explore the chemistry of the catalase enzyme (Gelasco, A., et al. Inorg. Chem., 1997, 36, 1829). The general structure of this complexes is the ligand binding to both manganese ions, with µ-O- alkoxide moieties from the ligand bridging the two manganese ions. The general structure of this family of complexes is shown by the crystal structure in Figure 2.
Figure 2. X-ray crystal structure of [Mn(III)(2-OHsalpn)]2. The ligand in this complex was prepared with 5-nitrosalicylaldehyde.
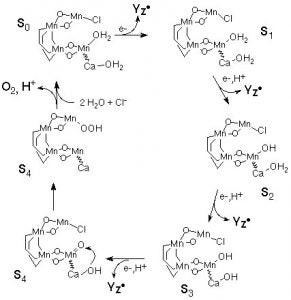
One of the key features of this system is that it is allows one to produce a series of complexes in four different overall oxidation states with only minor structural changes. These dimers have been prepared in the Mn(II)Mn(II), II/III, III/III, and III/IV oxidation states, a rare example of such a range of oxidation states with one ligand. This is key, since the catalase enzyme also exhibits this same set of oxidation states in its as isolated form. The catalase is considered to shuttle between the II/II and III/III oxidation states. When a one-electron process occurs to yield a II/III oxidation state, an ensuing two electron oxidation from the II/III state of the enzyme leads to the III/IV oxidation state, which is not active and only slowly over time is reduced back to a catalytically viable oxidation state. The [Mn(III)(2OHsalpn)]2 and [Mn(II)(2-OHsalpn)]2 complexes have been shown to be competentin the disproportionation of hydrogen peroxide, and that one is generated from the other if only turn over is allowed to occur. Thus the initial and final catalytic compounds have been established. Furthermore, this system exhibits saturation kinetic behavior. A catalytic cycle for this system is proposed in Scheme 2. (Gelasco, A., et al., J. Am . Chem. Soc., in press).
Scheme 2. A proposal for the catalytic cycle of Catalase based on the kinetics of hydrogen disproportionation of the two complexes.
While the reaction with hydrogen peroxide leads to disproportionation, the reaction with alkyl peroxides leads to the generation of free radicals. This reaction has been explored with t-butylhydroperoxide and the results of the reaction with cyclohexene indicated that this is a radical reaction. (Caudle, M. T., et. al. Inorg. Chem., 1996, 35, 3577).
Another unique feature of this system is its ability to bind an additional small molecule on one of the manganese ions, and to shift one of the bridging alkoxides into a terminal ligand status. Thus methanol and THF, for example, will bind to the Mn(III)Mn(III) complex, to form what is referred to as the unsymmetric dimer. This has been exploited to explore the binding of a hydroxide to the manganese and to probe such a complex spectroscopically. Again this may have implications for the OEC, and this family of complexes has been explored with respect to HBDE, as well (Caudle, M. T. and Pecoraro, V. L. J. Am. Chem. Soc., 1997, 119, 3415), since this dimer will bind a water molecule to form the unsymmetric dimer. The HBDE for the deprotonation of a water bound to manganese could then be obtained. Furthermore, in the case of hydroxide bound to the dimer, it was found that the presence of the hydroxide mediates the reduction potentials of the Mn(III)Mn(III) to the Mn(III)Mn(IV) dimer redox couple with respect to dimers without hydroxide bound, so that the Mn(III)Mn(IV) is more easily attained. Based on our newest results, and the H-atom abstraction hypothesis, we have completed a potential catalytic cycle of the OEC, which is presented in Scheme 3 (Pecoraro, V. L., et al. Pure and Applied Chemistry, Proceedings of the 8th International Conference on Bioinorganic Chemistry).
Scheme 3. A proposal for the S-state cycle of the OEC.
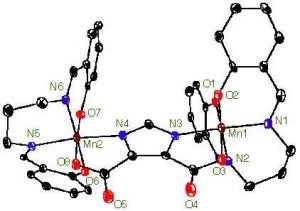
A new topic of research reapplies the H2salpn ligand in a new type of complex. It has been proposed that an imidazole bridge might exist in the OEC between two manganese ions. Our groups has been able to generate a dimeric complex bridged by an imidazole from the ligand 4,5-dicarboxyimidazole. A representative crystal structure is presented in Figure 3. Again we have been able to obtain this complex in several oxidation states. The Mn(IV)Mn(IV) oxidation state proved particularly unique, in that an unexpected EPR signal for a coupled Mn(IV)Mn(IV) system was observed (Caudle, M. T. et al. J. Am. Chem. Soc., 1997, 119, 9297).
Figure 3. X-ray crystal structure of [(Mn(IV)(3,5-di-t-busalpn)) 2DCBI]. The H2salpn ligand in this complex is derivatized with t-butyl groups in the 3 and 5 positions on the phenyl rings. They have been omitted for clarity.
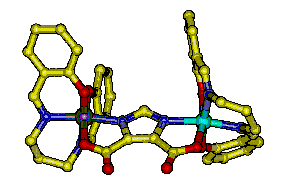
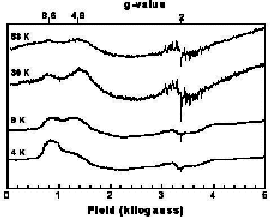
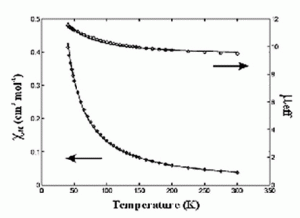
Significant attention has been focused on understanding the magnetic and electronic structure of manganese dimers as these compounds have been used extensively to model the spectroscopic signatures of the OEC. In all cases examined for [MnIIIMnIV(�2-O)2] 3+ the magnetic coupling has been antiferromagnetic and the ground state S=1/2. This has led to the observation of the classic g=2 multiline feature in the epr spectra of these complexes at cryogenic (4.2 K) temperatures. It has been speculated for some time that g=2 multiline features should be observed for excited states of ferromagnetically coupled [MnIIIMnIV]7+ centers. In order to test this hypothesis, we prepared Mn2(III/IV)(dtsalpn)2DCBI shown in the figure on the left. The incorporation of an imidazolate bridge causes a net ferromagnetic exchange coupling as shown in the plot of �eff vs. T. The coupling constant extracted from the fit of these data (J= + 1.4 cm-1) confirms this assignment. Having an authentic ferromagnetically coupled [MnIIIMnIV]7+ center, we obtained variable temperature X-band epr spectra of the compound in solution. We observe that the dominant low temperature feature is center at very low field at 4.2 K without discernable intensity at g=2 for a multilane component. Upon warming the sample to 30 K, a new g=2 feature develops which has the hallmark multilane feature. Thus, we have demonstrated the long sought after excited state, g=2 multiline signal. This work is published in J. Chem. Soc., ChemComm in 2003.
Arguably, second in importance to understanding the structure and chemistry of the S4 state of the OEC, is the definition of S0. This is because S0 is the product state of the reaction process. Based on XAS and EPR studies, a MnIIMnIIIMnIV2 ensemble has been proposed for the tetranuclear Mn cluster in S0. Such an oxidation state formulation is rare since it contains three different oxidation states. Under most circumstances, MnII and MnIV would comproportionate to make 2 MnIII. However, something special about the OEC appears to stabilize the different Mn oxidation states. We have prepared the first trinuclear Mn complex containing MnII and MnIV. The compound is Mn(II)Mn(IV)Mn(II)(pko)4(CH3O) 2(SCN)2.CH3OH , which is synthesized by the addition of 2,2′-dipyridyl etonoxime (Hpko) to a solution of sodium hydroxide and NaSCN in CH3OH, with MnCl2.4H2O. The neutral complex has 8 anions (2 thiocyanates, 2 µ-alkoxides and 4 pko– ligands), and the Mn oxidation state formulation of [Mn3]8+. There are two possibilities for the proper oxidation state assignment: [MnIIMnIII2]8+ or [MnII2MnIV]8+. We can confirm that the later composition is correct by comparing the properties of our new molecule to that of known trinuclear compounds of composition [MnIIMnIII2]8+. Key indicators supporting the proposed oxidation state assignment are the structures of the central octahedral MnIV ion and each MnII ion. The bond distances around the Mn(IV) atom average 1.91 E , and around each Mn(II) atom average 2.223 E. There is no evidence of Jahn-Teller distortion, which is typical of Mn(III).
Interestingly, this compound shows ferromagnetic exchange interactions. The exchange parameter between the central MnIVand terminal MnII is J= +6.13 cm-1 and g=2.09 . The magnetic ground state, confirmed by magnetization measurements at 4.5 K, is S=13/2 . The EPR spectral features of this compound are also different from [MnIIMnIII2]8+ trimers , as MnIIMnIVMnII has unstructured, low field transitions at 4.2 K while MnIIIMnIIMnIII exhibit broad features, with multiline character centered around 1600 G. Most important, we have been able to assess whether X-ray Absorption Spectroscopy can distinguish between the formulations [MnIIMnIII2]8+ and [MnII2MnIV]8+. The XANES spectrum for an authentic Mn(III)2Mn(II) trimer, is somewhat narrower than that seen for MnIIMnIVMnII or for its Cl- analog. When the XANES spectra for MnIIMnIVMnII were fit with a linear combination of MnII, MnIII, and MnIV, the best fits all had less than 15% MnIII, while the corresponding fits for MnIIIMnIIMnIII gave nearly 70% MnIII and never gave composition of MnIV > 15%. This work demonstrates that XANES should be able to discriminate the MnIIMnIIIMnIV2 composition for the OEC.
In conclusion, we have been able to apply a broad range of model complexes to broaden the understanding of the biological role of manganese. We have produced a variety of information that has provided new and exciting data for the field of bioinorganic manganese chemistry. Our research in the areas described above is ongoing, with many experiments left to be done. Thus, while we have accomplished much, there are still numerous frontiers left for our laboratory to explore in the context of the biological role of manganese.
Recent Project Publications (1995-present)
- Dissertations:
- Andrew Gelasco, 1995, “Low Valent Manganese Complexes as Functional Models for the Mn Catalases and the Alternate Catalase Reaction of the Oxygen Evolving Complex”.
- Neil A. Law, 1999, “Properties and Reactivity of Model Complexes for the Oxygen Evolving Complex of Photosystem II”.
- Wen-Yuan Hsieh, 2002 “Synthesis and Reactivity of the High-Valent Monomeric and Dimeric Manganese Complexes.”
- Journals, Periodicals, and Books:
- Gelasco, A.; Baldwin, M.; Pecoraro, V.L. “A Modelling Approach For Understanding the Mechanism of Manganese Enzymes” in Mechanistic Bioinorganic Chemistry, H.H. Thorp and V.L. Pecoraro, Eds., Adv. Chem Series, ACS Books, Washington, D.C. 1995, 265.
- Baldwin, M.J.; Kampf, J.W.; Kirk, M.L.; Pecoraro, V.L. “Structural and Magnetic Studies of Mn(II) Complexes of the Imidazole Containing Ligand 5-NO2-salimH [5-NO2-salimH2= 4-(2-(5-nitrosalicylideneamino)ethyl)imidazole] With Varying Nuclearity,” Inorg. Chem., 1995, 34, 5252.
- Baldwin, M.; Gelasco, A.; Pecoraro, V.L. “Modelling the Reactivity of the Oxygen Evolving Complex” in An Inorganic Perspective of Life, 1995,D.P. Kessissoglou, D. Coucouvanis and M. Kanatzidas, eds., Elsevier, Amsterdam, 287.
- Law, N.A.; Machonkin, T.E.; McGorman, J.P.; Larson, E.J.; Kampf, J.W.; Pecoraro, V.L. “A Structurally Characterized DiChloro-Mn(IV) Complex Capable of Chlorinating Alkenes,” J. Chem. Soc. Chem Comm., 1995, 2015.
- Pecoraro, V.L.; Baldwin, M.J.; Caudle, M.T.; Law, N.A.; Kampf, J.W.; Rasmussen, P.G. “A Modelling Approach for Understanding Water Oxidation in Photosynthesis,” Procedings of the Xth International Photosynthesis Congress, Kluwer P. M., ed; Academic Publishers: Dordrecht, The Netherland, 1995.
- Gelasco, A.; Askenas, A.; Pecoraro,V.L. “The Tetranuclear Complex [Mn(II)(picpn)]4 Efficiently Disproportionates Hydrogen Peroxide,” Inorg. Chem., 1996, 35, 1419.
- Caudle, M. T.; Riggs-Gelasco; P.; Gelasco,, A.K.; Penner-Hahn, J.E.; Pecoraro, V.L. “Homolytic Cleavage of tert-butyl hydroperoxide by Mn2(2-OHsalpn)2 to Form the One-Electron Oxidized Compound Mn2(2-OHsalpn)2 OH having a Terminal Hydroxo Ligand,” Inorg. Chem., 1996, 35, 3577.
- Baldwin, M.J.; Pecoraro, V.L. “Energetics of the Proton-Coupled Electron Transfer in High-Valent Systems: Models for Water Oxidation by Electron Transfer in High-Valent Systems: Models for Water Oxidation by the Oxygen Evolving Complex of Photosystem II” J. Am. Chem. Soc., 1996, 118, 11325.
- Caudle, M.T.; Kampf, J.W.; Kirk, M.L.; Rasmussen, P.G.; Pecoraro, V.L.; “The First Binuclear Mn(IV) Complex Containing a Bridging Imidazolate Ligand Exhibits Unique EPR Spectral Features”, J. Amer. Chem. Soc., 1997, 119, 9297-9298.
- Gelasco, A.; Kirk, M.L.; Kampf, J.W.; Pecoraro, V.L. “The [Mn-2(2-OHsalpn)2]2-,1-,0,1+ System: Synthesis, Structure, Spectroscopy and Magnetic Characterization of the First Dinuclear Manganese Series Containing Four Distinct Oxidation States” Inorg. Chem., 1997, 36, 1829.
- Caudle, M.T.; Pecoraro, V.L. “Hydrogen Atom Abstraction of Bound Substrate is a Viable Mechanism for Cluster Oxidation in the Oxygen Evolving Complex of Photosystem II,” J. Am. Chem. Soc., 1997, 119, 3415.
- Rompel, A.; Andrews, J.C.; Cinco, R.M.; Wemple, M.W.; Christou, G.; Law, N.A.; Pecoraro, V.L.; Sauer, K. Yachandra, V.K.; Klein, M.P. “Chlorine K-Edge X-ray Absorption Spectroscopy as a Prrobe of Chlorine-Metal Bonding: Manganese-Chloride Model Systems with Relevance to the Oxygen Evolving Complex in Photosystem II,” J. Am. Chem. Soc., 1997, 119, 4465.
- Randall, D.W.; Gelasco, A.; Caudle, M.T.; Pecoraro, V.L.; Britt, R.D. “1H ESE-ENDOR and ESEEM Characterization of Water and Methanol Ligation to a Dinuclear Mn(III)Mn(IV) Cluster,” J. Am. Chem. Soc., 1997, 119, 4481.
- Gelasco, A.; Bensiek, S.; Pecoraro, V.L.; “The[Mn2(2-OHsalpn)2]2-,1-,0 System: An Efficient Functional Model for the Reactivity and Inactivation of the Manganese Catalases”, Inorg. Chem., 1998, 3301-3309.
- Pecoraro, V. L.; Baldwin, M. J.; Caudle, M. T.; Hsieh, W.-Y.; Law, N. A.; “A Proposal For Water Oxidation in Photosystem II”, Pure & Appl. Chem., 1998, 70, 925-929.
- Law, Neil A.; Caudle, Tyler; Pecoraro, Vincent L.; “Manganese Redox Enzymes and Model Systems: Properties, structures, and Reactivity”, Advances in Inorganic Chemistry, Vol. 46, 1999, pp 305-440.
- Pecoraro, V.L. “A Modeling Approach for Understanding Photosynthetic Water Oxidation”; Photosyn. Resch., in Photosynthesis: Mechanisms and Effects,G. Garab (ed.), Kluwer Academic Publishers, Dordrecht 1998, 1247-52.
- Yocum, C.F.; Pecoraro, V.L.; “Recent Advances in the Understanding of the Biological Chemistry of Manganese”, Current Opinions in Biology, 1999, 3, 182-187.
- Pecoraro, V.L.; Hsieh, W-Y; “The Use of Model Complexes to Elucidate the Structure and Function of Manganese Redox Enzymes”, Chapter 14 in Metals in Biological Systems, Astrid Sigel and Helmut Sigel (eds.) Marcel-Dekker, Inc., Basel, Switzerland 2000, 37, 429-504
- Baldwin, M.J.; Law, N.A.; Stemmler, T.L.; Kampf, J.W.; Penner-Hahn, J.E.; Pecoraro, V.L.; “Reactivity of [{MnIV(salpn)}2(n-O,n-OCH3]+ and [{MnIV(salpn)} 2(n-O,n-OH)]+: Effects of Proton Lability and Hydrogen Bonding”, Inorg. Chem.,1999, 38, 4801-4809.
- Slebodnick, C.A.; Law, N.A.; Pecoraro, V.L. “A Comparison of Manganese and Vanadium Peroxidase, Haloperoxidase and Catalase Activities”; B. Meunier (ed.) (Imperial College Press: London), 2000, 215-67
- Law, N.A.; Kampf, J.W.; Pecoraro, V.L.; “A Magneto-Structural Correlation Between the Heisenberg Constant, J, and the Mn-O-Mn Angle in [MnIV(n-O)]2 Dimers”; Inorg. Chim. Acta., 2000, 297, 252-64 (a special volume in honor of Prof. Stephen J. Lippard).
- Caudle, M.T.; Pecoraro, V.L, “Mechanism for the Reduction of the Mixed-Valent Mn(III)Mn(IV)[2-Ohsalpn]+ Complex by Tertiary Amines”; Inorg. Chem., 2000, 39, 5831-5837.
- Peloquin, J.M.; Campbell, K.A.; Randall, D.W.; Evanchik, M.A.; Pecoraro, V.L.; Armstrong, W.A.; Britt, R.W. “55Mn ENDOR of the S2-state Multiline EPR Signal of Photosystem II: Implications on the Structure of the Tetranuclear Cluster”; J. Am. Chem. Soc. 2000, 122, 10926-10942.
- Rajendiran, T.M.; Kampf, J. W.; Pecoraro, V.L. “A Structurally Characterized Monomeric Mn(IV) Complex in a Discrete N2O4 Coordination Environment, ” Inorg. Chim Acta 2002, 339, 497-502 (Issue in honor of Helmut Sigel).
- Triller, M.U.; Hseih, W.-Y.; Pecoraro, V.L.; Rompel, A.; Krebs, B. “Preparation of Highly Efficiently Manganese Catalase Mimics,” Inorg. Chem., 2002, 41, 5544-5554.
- Hseih, W.-Y.; Pecoraro, V.L. “Oxidation of m-Chlorobenzoic acid by Mn(V)=O complexes,” Inorg. Chim Acta, 2002, 341, 113-117 (Special issue honoring K.N. Raymond).
- Rajendiran, T.M.; Kirk M.L.; Setyawati, I.; Caudle, M.T.; Kampf, J. W.; Pecoraro, V.L. “Isolation of the first ferromagnetically coupled Mn(III/IV) complex ” Chem Comm 2003 (7): 824-825
- Rajendiran, T.M.; Caudle, M.T.; Setyawati, I.; Kampf, J.W.; Pecoraro, V.L. “Evaluating Hydrogen Bond Interactions in Enzymes Containing Mn(III) -Histidine Complexation Using Manganese Imidazole Complexes,” JBIC 2003 8 (3): 283-293 FEB
- Alexiou, M.; Dendrinou-Samara, C.; Karagianni, A.;Biswas, S.; Zaleski, C.; Yoder, D.; Kampf, J.W.; Penner-Hahn, J.E.; Pecoraro, V.L.; Kessissoglou, D.P. “Models For the Lower S States of Photosystem II: A Trinuclear Mixed-Valent MnII/MnIV/MnII Complex,” Inorg. Chem, 2003, 42, 2185-2187.
- Maria Alexiou, Curtis M. Zaleski, Catherine Dendrinou-Samara, Jeff Kampf,Dimitris P. Kessissoglou, Vincent L. Pecoraro; “Trinuclear Mixed-Valent MnII/MnIV/MnII Complexes – Structure and Magnetic Behavior”Zeitschrift f|r anorganische und allgemeine Chemie,2003, 629(12-13),2348-2355.
- Triller M.U; Pursche D.; Hsieh W.Y.; Pecoraro V.L.; Rompel A.; Krebs B.; “Catalytic oxidation of 3,5-di-tert-butylcatechol by a series of mononuclear manganese complexes: Synthesis, structure, and kinetic investigation ” Inorg. Chem. , 2003 , 42(20)6274-6283.
- Weng, T.C., Hsieh, W-Y., Uffelman, E. S., Gordon-Wylie, S.W., Collins, T., Pecoraro, V.L. and Penner-Hahn, J.E.,”XANES Evidence Against a Manganyl Species in the S3 State of the Oxygen Evolving Complex”, J. Am. Chem. Soc., 2004, 126, 8070.
- Hsieh, W-Y., Campbell, K., Britt, R.D., Yoder, D., Penner-Hahn, J.E. and Pecoraro, V.L., “The First Spectroscopic Model for the S1 State Multiline Signal of the OEC”,Biochim. Biophys. Acta: Bioenergetics, 2004, 1655, 149.
- Angaridis, P.; Kampf, J. W.; Pecoraro, V. L.; “Multinuclear Fe(III) Complexes with Polydentate Ligands of the Family of Dicarboxyimidazoles: Nuclearity- and Topology-Controlled Syntheses and Magneto-Structural Correlations” Inorg. Chem. ,2005, 44(10), 3626-3635.
- Scarpellini, M.; Wu, A. J.; Kampf, J. W.; Pecoraro, V. L.; “Corroborative Models of the Cobalt(II) Inhibited Fe/Mn Superoxide Dismutases” Inorg. Chem. , 2005, 44(14), 5001-5010.
- Hsieh, W.Y.; Zaleski, C. M.; Pecoraro, V.L.; Liu, S. “Mn(II) Complexes of Monoanionic Bidentate Chelators: The x-ray Crystal Structures of Mn(DHA)2(CH3OH)2 (DHA=Dehydroacetic Acid) and [Mn(ema)2(H2O)]2 2H2O (Hema= 2-Ethyl-3-Hydroxy-4-Pyrone), Inorg. Chim. Acta, 2006, 359 (1), 228-236
- Gatjens, J.; Sjodin, M.; Pecoraro, V.L.; Un. S. “The relationship between the manganese(II) zero-field interaction and Mn(II)/Mn(III) redox potential of Mn(4′-X-terpy)2 complexes” J. Amer. Chem. Soc., 2007, 129, 13825.
- Signorella, S.; Buldt-Karentzopoulos, K.; Rompel, A.; Krebs, B.; Pecoraro, V.L.; Tuchagues, J.-P.; “Reevaluation of the Rates of Mn Catalase Mimics” Inorg. Chem., 2007, 46, 10864-10868.
- Mullins, C.S.; Pecoraro, V.L. “Reflections on Small Molecule Manganese Models that Seek to Mimic Photosynthetic Water Oxidation Chemistry,” Coord. Chem. Reviews, 2008, 252, 416-443.
- Sjodin, M.; Gatjens, J.; Tabares, L.C.; Thuery, P.; Pecoraro, V.L.; Un, S. “Tuning the Redox Properties of Manganese(II) and its Implications to the Electrochemistry of Manganese/Iron Superoxide Dismutases,” Inorg. Chem., 2008, 47, 2897-2908.
- Pecoraro, V.L.; Hseih, W.Y.; “In Search of High Valent Intermediates in Schiff Base Containing Manganese Dimers,” Inorg. Chem., 2008, 47, 1765-1778
- Meelich, K.; Zaleski, C.; Pecoraro, V.L.; “Using Small Molecule Complexes to Elucidate Features of Photosynthetic Water oxidation”, Phil. Trans. Royal Soc., 2008, 363, 1271-1281
- Gätjens, J.; Kampf, J.W.; Thuéry, P.; Pecoraro, V.L “Corroborative Cobalt and Zinc Models of α-amino-β-carboxymuconic-ε-semialdehyde decarboxylase (ACMSD),” Dalton Transactions, 2009, 51-62.
- Gätjens, J.; Kampf, J.W.; Pecoraro, V.L “Preparation of a new 16-MC-4 Structure Type That Captures Mn(II) in the Central Cavity” Inorg. Chim. Acta, 2009, 362, 878-886 [special issue in honor of Bernhard Lippert]. DOI:10.1016/j.ica.2008.02.006
- Ellis, P.D.; Lipton, A.S.; Sears, J.A.; Yang, P.; Dupuis, M.; Boron, III, T. T.; Pecoraro, V. L.; Stich, T.; Britt, R. D. “The 55Mn Solid State NMR Spectroscopy of bis(-oxo)dimangan-ese(IV) [Mn2O2(salpn)2]. An Antiferromagnetically Coupled Mn4+ Dimer at Cryogenic Temperatures” J. Amer. Chem. Soc. 2010, 132 (47), pp 16727–16729