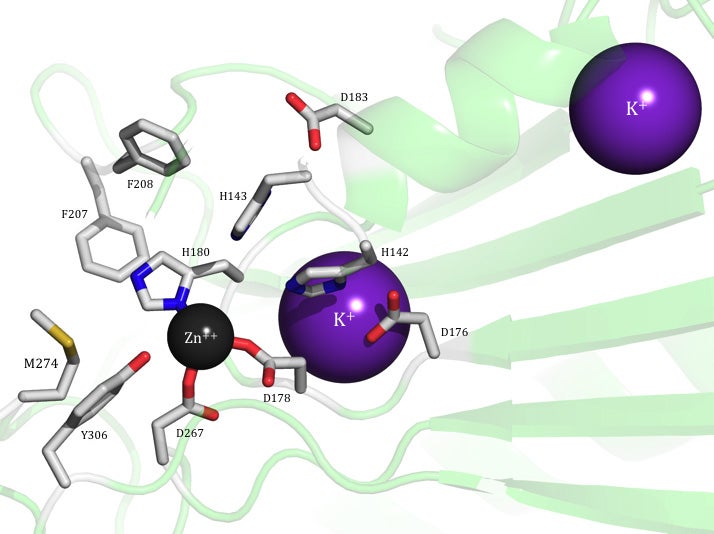
 Acetyl lysine deacetylases (acKDACs) catalyze the hydrolysis of N-acetyl lysine residues and play an important role in the regulation of gene expression. In the Fierke Lab, we seek to understand the function of such important enzymes in the cell, primarily looking at acKDAC8 and acKDAC11. Particularly, we work towards identifying novel substrates, understanding how they interact with other proteins and biomolecules in order to modulate their function, and determining their roles in different disease states. We use a variety of methods to answer these questions, including enzymatic assays of in vitro samples using acetylated peptides and proteins, western blots and proteomics of mammalian cell culture samples to investigate cellular behavior, single molecule fluorescence and photo crosslinking approaches to visualize binding partners, and a variety of other biochemical and chemical biology tools.
Acetyl lysine deacetylases (acKDACs) catalyze the hydrolysis of N-acetyl lysine residues and play an important role in the regulation of gene expression. In the Fierke Lab, we seek to understand the function of such important enzymes in the cell, primarily looking at acKDAC8 and acKDAC11. Particularly, we work towards identifying novel substrates, understanding how they interact with other proteins and biomolecules in order to modulate their function, and determining their roles in different disease states. We use a variety of methods to answer these questions, including enzymatic assays of in vitro samples using acetylated peptides and proteins, western blots and proteomics of mammalian cell culture samples to investigate cellular behavior, single molecule fluorescence and photo crosslinking approaches to visualize binding partners, and a variety of other biochemical and chemical biology tools.
Currently, we are utilizing the incorporation of photo reactive, non-natural amino acid – p-benzonyl-4-phenylalanine (pBpa) – into acKDAC8 as a way to trap both substrates and binding partners, and possibly know how this enzyme is regulated in vivo. We are also working on the development a chip-based proteomics method for screening full-length protein substrates. A variety of methods are used to validate hits from these proteome chips, including computational prediction algorithms, determination of in vitro kinetic parameters on acetylated peptides, and expression of singly-acetylated recombinant proteins for analysis. With acKDAC11, the most recently identified isozyme, our work is more focused on basic enzyme characterization methods. We’re investigating substrate selectivity, the potency of common inhibitors, and the effects of potential PTMs on acKDAC11 itself.
Collaborators on this project:
Ora Furman at The Hebrew University of Jerusalem
Andrew Andrews at Fox Chase Cancer Center
Geeta Narlikar at The University of California, San Francisco
John Denu at The University of Wisconsin
Phillip Cole at Johns Hopkins University
Brent Martin at The University of Michigan

