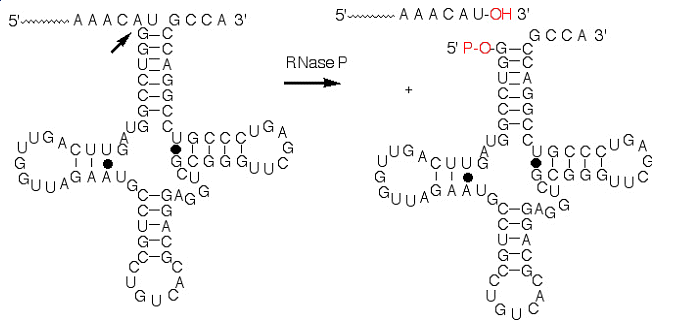
The long term goal of this research project is to further our understanding of (1) the mechanisms of catalysis used by ribozymes as compared to protein enzymes, and (2) the structure of RNA binding proteins and protein/RNA complexes. To this end, we have begun investigating the enzymology and structural biology of the catalytic ribonucleoprotein RNase P. This novel catalyst contains an RNA subunit (P RNA) and a protein subunit (P protein). As of date, we solved the first structure of the protein component of ribonuclease P in a collaboration with the Christianson laboratory at UPenn and demonstrated that one of the functions of this protein is to bind the leader sequence of the precursor-tRNA substrate. This function places the protein component in the proximity of the active site and suggests that the active site occurs at the RNA/protein interface. This is the first case where the RNA and protein components both contribute to the molecular recognition properties of an enzyme. Furthermore, the positions of key metal ions have been identified using phosphorothioate substitution studies.
 Fierke Research Group
Fierke Research Group
Faculty, Lab, and Research Template Site