April 2024
Our first paper on C. elegans telomeres out in PNAS! Shilpa Padmanaban as a Ph.D. student in the lab showed how C. elegans telomeric ds DNA-binding proteins TEBP-1 and TEBP-2 adapted a prototypical DNA-binding domain to not only perform the canonical function but to also dimerize – a characteristic of telomeric ds DNA-binding proteins. Another great collaboration with Dr. Hiroki Shibuya’s lab, which performed the in vivo validation. www.pnas.org/doi/10.1073/pnas.2316651121
August 2023
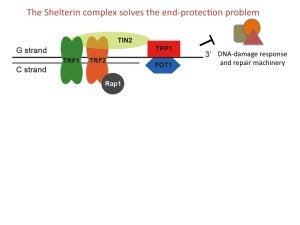
A HUGE moment for the lab! Our discovery of the POT-hole of POT1 as the structural mechanism for protecting the 5′ end at the double-stranded-single-stranded junction of our chromosome ends is out in Science! Congratulations to Valerie Tesmer and Kirsten Brenner.
Video starts with the previously solved structure of human POT1 with ss DNA (PDB: 1XJV) and then transitions into the new structure of the protein bound to the telomeric ds-ss DNA junction. The 5′ phosphate is shown as spheres in CPK coloring.
Free download:
https://www.science.org/stoken/author-tokens/ST-1380/full
normal journal link: https://t.co/ozZIuZrTsh
This discovery opens so many avenues for the lab and raises so many interesting questions of fundamental and clinical relevance!!!
July 2023
Congratulations to Shilpa Padmanaban and Ritvija Agrawal for successfully defending their Ph.D.!!! We are so proud of you and sad that you will be leaving us but also very happy to see you go on to do bigger things!
March 2023
Congratulations to Shilpa Padmanaban for publishing a first author paper in Life Science Alliance! This is a tour de force in which Shilpa used a combination of site-directed mutagenesis, domain deletions, and a combination of microscopy and classical enzymology to reveal several new elements within TERT that are important for recruitment to telomeres and catalytic function. https://www.life-science-alliance.org/content/6/6/e202201727
September 2022
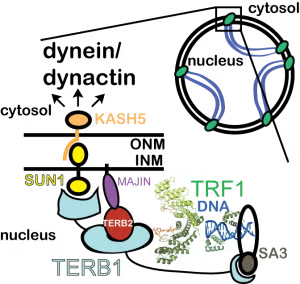
Happy to report that our NIH R01 grant from the NICHD was funded for the full five years! These funds will be critical in supporting a collaborative project (with the labs of Drs. Morgan DeSantis, Hiroki Shibuya, and Allen Liu) to understand how the SUN1-KASH5 LINC complex helps move chromosomes during meiosis.
June 2022
A review by Kirsten Brenner from our lab on the consequences of replication stress at telomeres and how DNA repair pathways acting at telomeres can contribute to genomic instability is now published in Trends in Biochemical Sciences. This was ALL KIRSTEN and is a testament to her outstanding critical thinking and writing skills.
https://www.sciencedirect.com/science/article/pii/S096800042200072X
June 2022
Congratulations to MCDB Ph.D. student Shilpa Padmanaban for winning the Okkelberg Ph.D. Fellowship.
June 2022
Congratulations to Jonathan Williams, an MSTP student in the lab, for being awarded a fellowship through the NIH T32 grant of the CMB program.
June 2022
Congratulations to Ritvija Agrawal on her eLife paper! Ritvija did this work along with co-mentorship of Dr. Morgan DeSantis and collaboration on the in vivo aspects with Dr. Hiroki Shibuya’s lab. In this work, we showed how the meiosis-specific LINC protein KASH5 binds dynein and activates dynein to move chromosomes along the nuclear envelope. Check it out at https://t.co/FlOX27i0k1
October 2021
Congratulations to MSTP student Jackie Graniel and our wonderful collaborators Ivan Maillard (UPenn), Katy Keegan (UM), and Sue Hammoud (UM) and their labs for our latest paper that was published in Life Science Alliance. Jackie and her co-authors characterized the hematopoietic and germline compartments of a mouse knocked in for a telomeropathy mutation in the telomere protein TPP1 to find that the mutation spares hematopoiesis but results in a progressive loss of fertility. Our studies suggest that humans and mice possess distinct vulnerabilities towards telomere shortening. While the primary vulnerability in humans lies in the soma, in mice it lies in the germline. Check it out at https://www.life-science-alliance.org/content/5/1/e202101208
August 2021
Our structure of a meiosis-specific BRCA2 complex containing the MEILB2 protein was published in Nature Structural & Molecular Biology. Congratulations to graduate students Devon Pendlebury and Ritvija Agrawal for the crystallographic and biochemical efforts, and a big thank you to our longstanding collaborators in the Hiroki Shibuya lab in Sweden.
https://www.nature.com/articles/s41594-021-00635-0
June 2021
Congratulations to Cassie for passing her checkpoint 2 exam! What a smooth sail. Well done, Cassie.
April 2021
Huge Congratulations to Ritvija Agrawal for being awarded the American Heart Association Predoctoral Fellowship for two years! She is the first trainee to win an external fellowship in the lab! Well done, Ritvija!
October 2020
Huge Congratulations to MSTP student in the lab Jackie Graniel for winning the best oral presentation in Cell/Molecular Biology at the recent SACNAS conference! Way to go, Jackie! Here is the announcement:
STEM Students Recognized for their Research and Presentation Skills at 2020 SACNAS – The National Diversity in STEM Conference
August 2020
Congratulations to MCDB Ph.D. student Ritvija Agrawal for winning the CEW+ (CENTER FOR THE EDUCATION OF WOMEN +) Scholarship Award! She was named a Sarah Winans Newman Scholar for this year.
August 2020
Congratulations to MSTP student Jackie Graniel for being awarded the Rackham Student Ally Diversity Grant (Cell and Developmental Biology Department) https://rackham.umich.edu/faculty-and-staff/faculty-and-program-funding/rackham-faculty-allies-and-student-ally-diversity-grants/
This adds to several other accomplishments of Jackie in the area of enhancing DEI on campus and in our community, including actively participating in the Development of Future Biologists (DFB) teaching program, and founding and leading the SACNAS chapter of the University of Michigan.
December 2019
Congratulations to Valerie, Eric and the rest of the crew for a recently published paper in PNAS where we reveal specific regions in human telomerase that mediate an interaction with TPP1 at telomeres.
https://www.pnas.org/content/early/2019/12/09/1911912116
July 2019
Sheri Grill defends her thesis in style! First MCDB PhD graduate from the lab. What a wonderful job, Sheri. We are all proud of you and look forward to all the great achievements that lie ahead of you in the future.
June 2019
Shilpa and Ritvija prevail over prelims Checkpoint 2! Congratulations to both of them.
June 2019
Sheri’s paper is (finally) out in Cell Reports!
Its the long and short of TPP1 protein that you need to know about…
https://www.sciencedirect.com/science/article/pii/S2211124719307028?via%3Dihub
March 2019
Devon Pendlebury, 4th year Chemical Biology PhD student was awarded a Rackham Predoctoral Fellowship. Congratulations, Devon!
October 2018
MSTP student Jackie Graniel is now funded by the Biology of Aging Training Program (NIH T32).
Congratulations, Jackie!
August 2018.
Eric Smith defended his PhD thesis successfully. Congratulations, Eric!
December 2017
Our work uncovering a second telomerase-interacting surface within TPP1 was accepted for publication in Cell Reports.
Congratulations to Sheri and Valerie!
October 2017
Our work describing how telomeres attach to the inner-nuclear membrane during meiosis came out online in Nature Structural & Molecular Biology.
Congratulations to Devon, Valerie, and Eric!
https://www.nature.com/articles/nsmb.3493
May 2017
Our paper on the development of an inducible Cas9 system for editing of genes in cultured human cells is now out in Analytical Biochemistry. Congratulations to Kamlesh, Sheri, and Jackie!
http://www.sciencedirect.com/science/article/pii/S0003269717301902
April 2017
Honors student Jonathan Williams was awarded the Marshall Nirenberg Life Sciences award (Goldstein Honors Prize) that is given to the top life sciences honors graduating student by the Literature Science and Arts College of UM.
April 2017
We have been awarded a Research Scholar Grant by the American Cancer Society
November 2016
Congratulations to Devon Pendlebury for passing her Ph. D. candidacy examination!
Link to our PNAS paper on a DC mutation in the telomere protein TPP1
http://www.pnas.org/content/113/46/13021.long
Congratulations to second year Program in Chemical Biology graduate student Devon Pendlebury for been awarded the Rackham International Student Fellowship Award!
October 2016
Congratulations to second year Program in Chemical Biology graduate student Devon Pendlebury for been awarded the Rackham Graduate Student Research Grant.
September 2016
Another paper from the lab accepted this month, this time in PNAS! This is ALL US… More details soon.
September 2016
Congratulations to Eric! He was awarded a Biology of Aging Training Grant Fellowship for his structural studies on the telomeric protein TPP1.
September 2016
A paper to which our lab contributed to has been accepted for publication in Oncogene. More details soon.
August 2016
Congratulations to the lab! We were awarded an R01 by the NIH (NIGMS) for understanding the function and regulation of the TPP1 protein in human cells.
May 2016
A review written by our collaborators (Labs of Ivan Maillard and Katy Keegan @ UM) and us was published in Journal of Clinical Investigation. It details the role of shelterin in preventing DNA damage signaling at telomeres, telomerase function, and hematopoiesis.
April 2016
Congratulations to Jonathan Williams (undergraduate researcher in the lab) for winning the Otto Graf Scholarship, and also for receiving an Honorable Mention in the nationwide Barry Goldwater Scholarship competition.
October 2014
The first paper from the lab was published in the October 1st issue of Genes & Development. Here we showed that a single amino acid deletion in the TEL patch of TPP1 results in reduced recruitment of telomerase to telomeres, reduced telomerase processivity, and ultimately to a severe case of the telomere-dysfunction disease dyskeratosis congenita. The TPP1 mutation in the proband was identified via whole exome sequencing by a team of scientists at the NCI of NIH, led by Dr. Sharon Savage. Our previous studies have highlighted the role of the TEL patch of TPP1 in telomerase function with regards to cancer cell proliferation. The current study confirms that the TEL patch is also critical for the self-renewal of stem cells, an aspect that is compromised in patients with dyskeratosis congenita. The link to the table of contents on this G&D issue is:
http://genesdev.cshlp.org/content/current
September 2014
Congratulations to Sheri for being awarded a Genetics Training Program fellowship for two years!