Many reports have indicated that the overall charge of the ions observed in nano-ESI experiments can have a significant effect on the information content of the IM-MS dataset obtained. For example, it has been observed that the quaternary structure of protein complexes is altered from its native form in more highly-charged ions in some cases. Also, evidence from collision induced dissociation (CID) has recently revealed dramatic charge-dependent mechanisms for product ion generation. The utility associated with charge manipulation is clear and we are in the process of critically evaluating a number of charge reduction approaches adapted for IM-MS analysis of protein complexes.
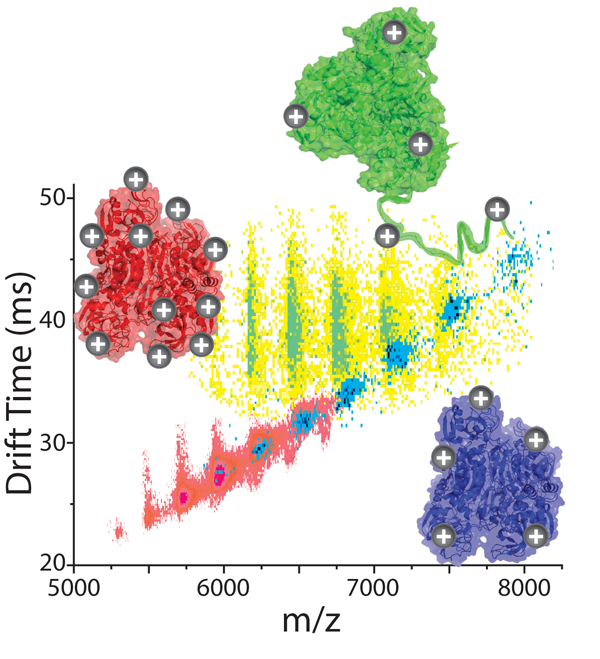
For more details, please refer to Bornschein, R. E., et al. Analyst., 2015, 140 (20).
For a detailed summary of current work in this research area, please visit the publication site.
- Welcome Addison, Hanyu, and Ryan!
- Congratulations to Iliana!
- Congratulations to Dr. Rojas Ramirez and Dr. Parson!
- Congratulations to Dr. Han!
- New Publication from the Ruotolo Lab Featured in UM News!
Contact
Ruotolo Group
University of Michigan
Department of Chemistry
Room 4550
930 N. University Ave, Ann Arbor, MI 48109-1055
Phone: (+1) 734-763-2443
Fax: (1+) 734-615-3718
